
Sem-O® (Semaglutide) is an oral glucagon-like peptide-1 receptor agonist (GLP-1 RA) formulated with SNAC® absorption technology to enable systemic exposure following gastric absorption.
It provides sustained GLP-1 receptor activation, supporting glucose-dependent insulin secretion, glucagon suppression, and improved glycemic control in adults with type 2 diabetes mellitus, offering a clinically validated non-injectable incretin-based therapy option.
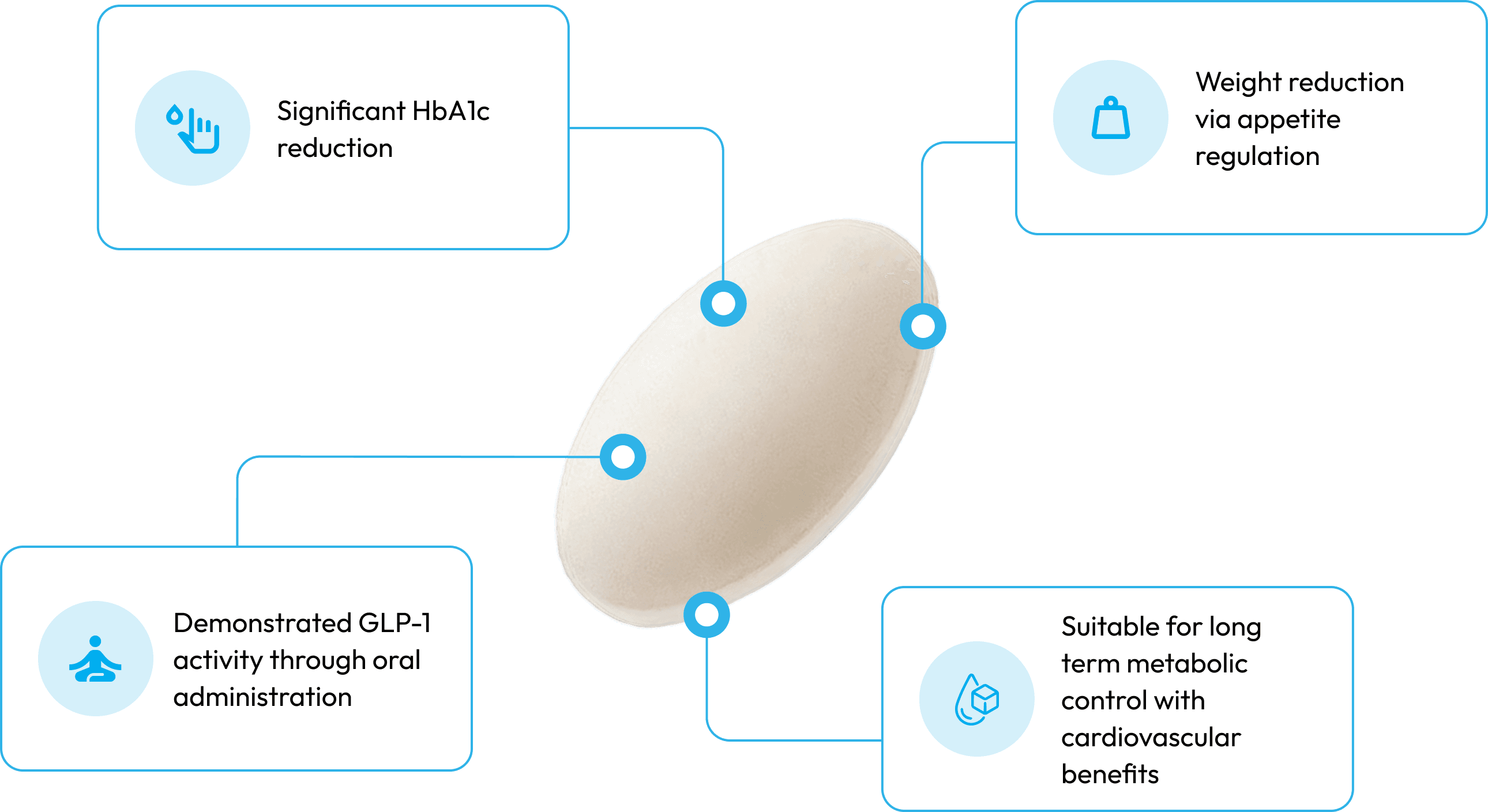
Key Features


SEM-O is a GLP-1 receptor agonist, with oral absorption enabled by SNAC technology.


Promotes insulin release in response to elevated glucose levels. [1]

Reduces excess glucagon to limit hepatic glucose output. [1]

Modulates appetite regulation to support reduced energy intake.
Your BMI
Enter your height and weight to see your Body Mass Index (BMI)
Use this calculator to check your patient’s body mass index (BMI), which can help you know your weight category.
Clinical studies demonstrate the efficacy of oral semaglutide. [2]
Browse clinical and real-world studies.
March 27, 2024
March 27, 2024
Your patient can take Sem-O exactly as per your instructions. The patient swallows one tablet whole once a day, with a sip of plain water (no more than 120 ml), on an empty stomach in the morning. Do not split, crush, or chew the tablet. Avoid taking it with any other liquids. Wait at least 30 minutes before eating, drinking, or taking other medicines. If the patient misses a dose, skip it and take the next dose at the regular time.
Like all medications, GLP-1 treatments can cause side effects. The most common are mild nausea, digestive changes, and decreased appetite, which typically improve after the first few weeks.
Women should stop taking Sem-O at least 2 months before planning pregnancy. Breastfeeding is not recommended during treatment with Sem-O.
The recommended starting dose of Sem-O is 3 mg once daily for the first 4 weeks. After that, the dose is increased to 7 mg once daily from weeks 5 to 8, and then to 14 mg once daily from week 9 onward, or as per your advice to your patients.
Semaglutide is contraindicated in patients with:
[1] Ref: Rev Endocr Metab Disord. 2022 Jan 7;23(3):521–539]
[2] http://2.https//myendoconsult.com/learn/the-ominous-octet-of-t2dm-m-o-a/#t-1760594955866